Vaccine Track Resources

Adult Vaccines - FY 2024H1 Update
Nationwide, average monthly adult vaccination claims (excluding flu, RSV, and COVID-19) increased 10% from Q1 to Q2 2024.
Read more
Adult Vaccines - FY 2023 Update
Data from Vaccine Track for the full year of 2023 continue to show a broad upward trend in adult vaccinations compared to the years immediately following the pandemic, suggesting that soon, the number of vaccination claims could consistently equal pre-pandemic levels. In 2023, overall adult vaccination claims were 7% lower than the claims in 2019, whereas vaccination claims in 2022 were nearly 15% lower than in 2019. Of note, for the first three quarters of 2023, vaccination claims exceeded 2019 levels by 1%.
Read more
Adult Vaccines - H1 2023 Update
Vaccinations continued to increase from January through June of 2023 as compared to the same time last year. In comparison to pre-pandemic levels, this marks the first time there has been an increase in H1 national immunization claims.
Read moreNational Immunization Awareness Month Update
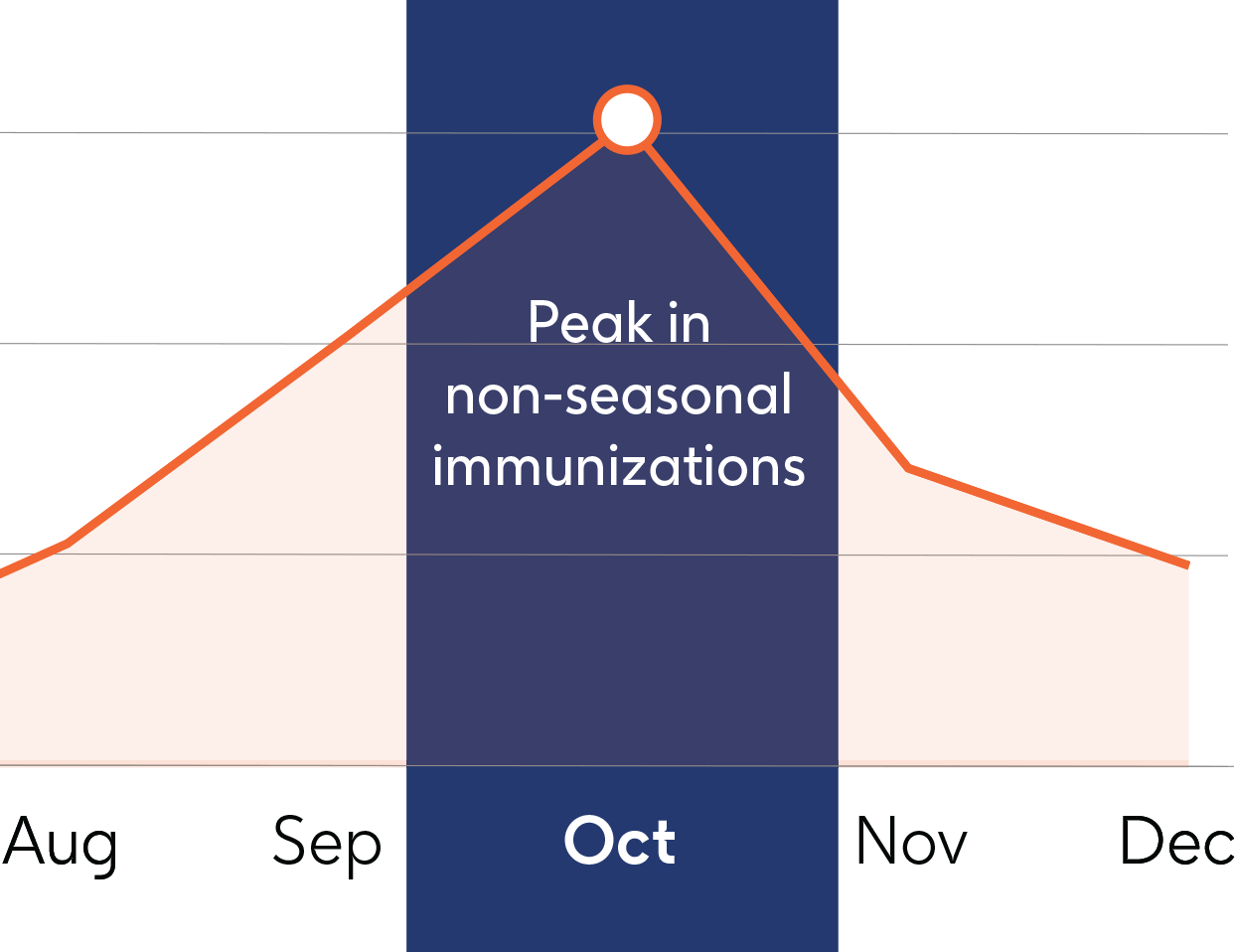
August is National Immunization Awareness Month (NIAM), which emphasizes the importance of routine immunization for people of all ages. This year, NIAM also highlights additional trends to watch for, including the implications of the end of the COVID-19 public health emergency, the expansion of full vaccine coverage without cost sharing for Medicare and Medicaid beneficiaries, and the FDA approval of new vaccines.
Read moreOptimizing Adult Immunizations in the U.S.
This white paper from CapView Strategies outlines the current vaccine coverage landscape in the US and offers tangible solutions for addressing adult immunization gaps. The paper reviews coverage of recommended vaccines in the U.S. since the passage of the Inflation Reduction Act, including expansions in vaccine coverage for adults in Medicare and Medicaid.
Read moreAdult Vaccines - Outlook for 2023
The COVID-19 pandemic had a significant impact on decreasing routine immunization rates both for children and adults — but it also provided lessons learned on how to address equity issues and system barriers that can be leveraged moving forward to improve rates. This is especially important to protect against respiratory diseases as we saw this year with the “tripledemic.”
Read more